Penetration, absorption and bio-equivalence:
VitroScreen’s expertise and capabilities

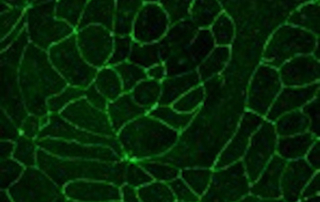
VitroScreen’s expertise has been established during almost 20 years and it is based on non-animal methods. VitroScreen performs pre-clinical studies to assess skin and epithelial absorption and penetration with green and HPLC-MS/MS, UPLC analytical techniques without using radiolabeled substances.
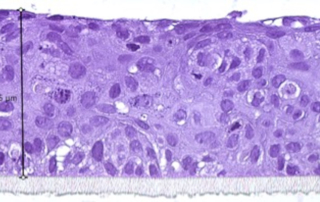
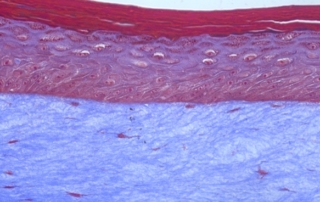
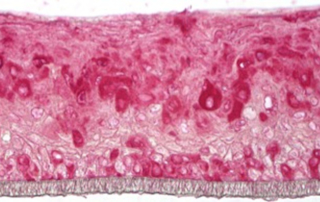
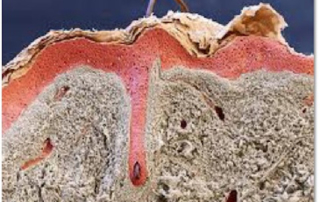
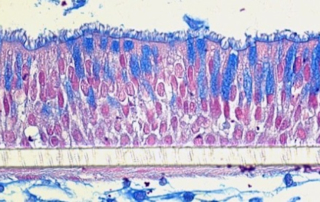
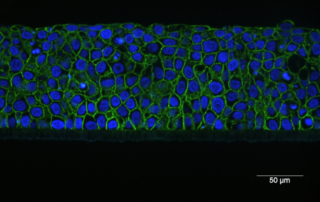
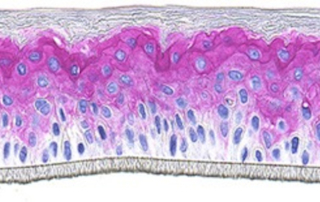
Absorption procedures have been internally validated with reference molecules on a wide range of biological models (human skin explants and 3D human reconstructed tissues). By using 3D human tissue models we can cover almost all body barriers: from intact or injured skin to different epithelia.
These studies allow to investigate local distribution at the site of application and are applicable to API, medical devices, chemicals, biocides and pesticides.
APPLICATIONS:
- Regulatory studies in GLP according to OECD TG 428 requirements, EFSA, EMA and FDA guidelines
- Bio-equivalence studies
- Characterization and regulatory classification of Medical Devices according to Rule 21 of MDR
- Drug Delivery and Controlled Release projects
- Absorption, Distribution, Metabolism and Excretion (ADME)
- Metabolites toxicokinetic on fully viable tissues
- Independent toxicology statement on penetration studies results (within the In Vitro Consultancy Unit)
OECD TG 428 as reference guideline
The in vitro method for skin absorption (OECD TG 428) introduces quality criteria and technical requirements that have to be satisfied to guarantee the validity of the study itself. VitroScreen applies these criteria to all penetration studies performed on skin explants or 3D organotypic tissue models, including skin, oral, vaginal, corneal, bladder, colorectal, nasal, intestinal tissues.
The possibility to assess the local distribution of functional ingredients at the site of application is particularly interesting for the medical device sector where ADME information requirements have been introduced for safety and classification purposes of substance-based medical devices (EU Regulation 2017/745, Rule 21). For this sector, but not limited to it, VitroScreen has developed specific protocols for absorption studies on intact and injured epithelia and mucosae.
In 2016 FDA introduced the possibility to perform in vitro permeation test to demonstrate the bio-equivalence of dermatological products, providing evidences of the relevance of the in vitro approach.