FUNDAMENTALS OF SKIN SENSITIZATION:
THE NEED FOR AN INTEGRATED APPROACH
The complexity of biological mechanisms described by the four identified Key Events (KEs), coupled with the mechanisms of a full validation process, makes the skin sensitization a very challenging regulatory endpoint.
To support the classification requirements (namely, REACH Regulation for neat chemicals and mixtures; Regulation 1223/2009 and SCCS notes of guidance for cosmetic products and ingredients; Regulation 2017/745 and ISO 10993-1 for Medical Devices) the newly established VitroScreen’s In Vitro Consultancy Unit is committed to provide customers with a comprehensive and integrated strategy to better address skin sensitization hazard and risk assessment.
The In Vitro Consultancy Unit supports Customers in:
- establishing the toxicological profile of ingredients
- providing an independent toxicological expert judgment (ERT toxicologist) on available data
- performing a gap analysis, if applicable
- identifying the most appropriate and sustainable experimental approach according to product type, chemical-physical properties and product category.
The In Vitro Consultancy Unit applies the rationale established within the Integrated Approaches to Testing and Assessment (OECD Guidance Document N. 256); in case of specific ingredients, mixtures or finished products, customized approaches are proposed.
VitroScreen performs a large set of validated OECD TGs for skin sensitization classification as well as some of those that are under validation: very often their specific features represent an important advantage to better address the regulatory needs by covering KE1, KE2 and KE3.
| TEST METHOD | KE | |
| Skin Sensitization | DPRA by LC–MS/MS | KE1 |
| KeratinoSensTM (OECD TG 442D) | KE2 | |
| EpiCS® in vitro skin sensitization test SkinInVitro Protocol, January 2020 |
KE2 | |
| Human-cell line activation test (h-CLAT) (OECD TG 442E) (standard and modified protocol) |
KE3 | |
| GARD® (Partnered)
• GARD®skin |
KE3 |
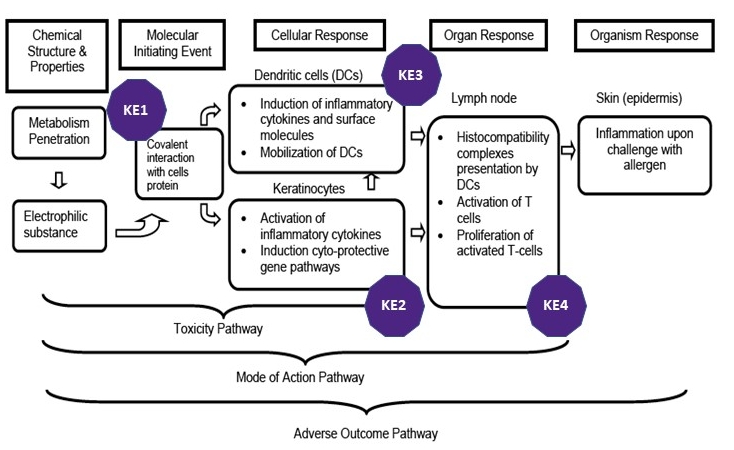
Image from ENV/JM/MONO(2012)10/PART1
Current regulatory status
The current knowledge of the chemical and biological mechanisms associated with skin sensitization has prompted the development of alternative (in silico, in chemico, in vitro) methods addressing specific key events (KEs) of the Adverse Outcome Pathway (AOP) process (ENV/JM/MONO(2012)10/ PART1 and PART2). Many of these methods have received international acceptance as OECD Testing Guidelines (TGs); others are under development and validation to extend the applicability domain of non-animal testing strategies to low soluble chemicals, complex mixtures and surfactants.